European Pharmacopoeia EP 2.2.44 and Total Organic Carbon
Abstract
The new revision of the European Pharmacopoeia chapter on Water for Injection (WFI) now permits the generation of WFI using Reverse Osmosis1 (RO), paving the way for companies to reduce their energy bills and carbon footprint by moving away from using stills. This paper discusses the increased focus on Total Organic Carbon (TOC) and conductivity measurement, especially for companies seeking to use RO water treatment plants to create WFI, and provides best-practice advice for calibration.
Background
In March 2016, the European Directorate adopted their new pharmacopoeia chapter on Water for Injection (WFI) after consultation with a working group of experts from the pharmaceutical industry. The previous form of this chapter instructed that WFI should be made from a still whereas in this new chapter, WFI production from reverse osmosis (RO) is permitted, provided that the water quality is equivalent to that produced by still.
However, during the consultation period, concerns were raised as to the robustness of RO compared to stills and the potential for microbial break-through, biofilm build up or for contamination from microbial bi-products. The increased importance of monitoring for conductivity and TOC were emphasised during the EDQM Expert Workshop2 as indicators of potential microbial contamination or from microbial by-products.
TOC and Conductivity
TOC analysis is a non-specific test, i.e. it is simply a measure of the carbon found in any organic compound in the water, it cannot tell you what type of organic molecule is present. A pharmaceutical-grade TOC analyser uses ultra-violet light (UV) to oxidise the organic molecules to release the carbon atoms present and then measures the difference in water conductivity caused by the resultant carbon dioxide.
TOC is to be calculated by measuring Total Inorganic Carbon (TIC) and Total Carbon (TC) and subtracting one from the other.
TC – TIC = TOC
Figure 2. TIC and TC are measured and TOC is calculated
The European Pharmacopoeia chapter on TOC for PW and WFI, EP 2.2.44, calls for complete oxidation of the organic molecule for accurate TOC analysis, i.e. if some of the carbon atoms are not oxidised and remain bound into the organic molecule, then they would not be measured and the TOC analyser would under-report TOC. For this reason it is important that the TOC analyser is capable of detecting when oxidation is complete before reporting TOC levels.
TOC results are reported in Parts Per Billion (ppb) which in this case is the mass (weight) of organic carbon per litre of water reported in mg/Litre. Longer-chain complex organic molecules may contain more carbon atoms than shortchain organic molecules, so equivalent numbers of the long- and short-chain molecules will be reported differently by the TOC analyser, with reported TOC from the long-chain organics delivering higher TOC results.
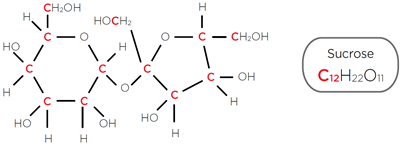
Figure 3. Organic molecule sucrose contains 12 carbon atoms
TOC analysers that use conductivity to measure the organic carbon can also be used to detect inorganic contamination. However, the pharmacopoeias are very specific regarding calibration of conductivity analysers and the TOC analyser would need to comply fully with their requirements. USP<645> is very clear that the conductivity meter cell constant must be verified against a certified conductivity solution and that the meter electronics must be verified by the use of certified calibration resistor.
Importance of TOC & Conductivity with the new EP chapter on WFI
The Working Party who supported the European Directorate warned that RO water treatment may not be as robust as a still and highlighted the role of TOC and Conductivity analysers as early detectors of potential impending microbial or endotoxin excursion events.

Figure 1. Working group supporting European Directorate emphasises the importance of TOC and Conductivity analysers
An increase in TOC results in a water system that usually has consistent TOC results can be an important indicator that either the quantity of organic molecules present in the water is increasing, or that there is a different, more complex long-chain molecule entering the water system. Such changes in TOC results can be an indicator that the integrity of the water treatment system is being threatened and can be an early warning of a potential impending microbial breech. As such events are often transient in nature, it is important to capture a sample of the water at the time when the TOC increase is detected so that it can be sent to the quality control laboratory for further analysis to try to get to the root cause of the transient.
Clearly TOC and Conductivity analysers must be calibrated correctly and their calibration validated to ensure that they are working correctly in case a change in the water chemistry occurs. Equally, the intensity of the oxidising UV lamp needs to be monitored to ensure that it is strong enough to oxidise organic molecules sufficiently to extract all of the carbon atoms for accurate measurement. Although TOC analysers cannot determine what type of organic molecules are present in the water, they can be used to detect a change in the organic chemistry in the water as longer-chain organic molecules tend to exhibit different oxidation profiles compared to short-chain organic molecules. If the TOC analyser is able to detect a change in oxidation profile when the water organic chemistry changes, this information combined with a change in TOC results can indicate to the user the need to carry out an investigation to determine if the water system integrity is potentially about to be compromised.
TOC and Conductivity Analysers: best practices for calibration
Calibration
Typically, most pharmaceutical manufacturers have their TOC analysers calibrated on a six-monthly basis. There are two types of calibration; calibration validation and calibration adjustment.
For calibration validation, certified calibration standards are run as grab samples and the performance of the TOC analyser is compared to the certified values. A ‘Pass’ is given if the reported values from the TOC analyser are within an acceptable percentage of the certified values of the standards (blank subtracted).
For calibration adjustment, certified calibration standards are run through the TOC analyser. The TOC analyser adjusts its calibration slope to give a best fit against the certified values of the standards. As long as the TOC analyser is able to change its slope to give a correlation coefficient (linearity factor) ≥0.990 and as long as the change in the calibration slope of the TOC analyser is not a large change from the original factory calibration, then the new calibration is considered acceptable and a ‘Pass’ is given. A change in the calibration slope that is a large deviation from the factory slope is considered an indication that something has gone wrong, i.e. either the analyser is not working correctly, or the calibration standards are not matching their certified value.
Although not mandated, it is considered best practice to carry out the following step-by-step procedure when calibrating a TOC analyser:
A. Carry out an ‘as found’ calibration validation before any adjustments are made. This confirms that the TOC analyser has been working within specification since the last calibration and that nothing has gone wrong since then.
B. Carry out a calibration adjustment. This ‘fine tunes’ the calibration of the TOC analyser to make it as accurate as possible and protects against gradual drift in calibration over extended periods of time
C. Carry out an ‘as left’ calibration validation. This confirms that the calibration slope adjustment made in step b) has resulted in an accurate calibration compared to traceable, certified standards
System Suitability
This test is designed to ensure that the TOC analyser is capable of analysing equally the range of organic contaminants that might occur in PW and WFI. This takes on a particular significance with the concerns around potential microbial contamination or from microbial by-products as the type of organic contamination may change with time.
Conductivity
The weakness of traditional conductivity instruments is that the measurement cell is inside the WFI pipe and inaccessible so the cell constant cannot be checked as recommended in the United States and European pharmacopoeias (USP and EP)3,4 using a conductivity solution of a known, certified value. TOC analysers that use direct conductivity measurement as part of their TOC analysis such as the PAT700 from Beckman Coulter can have the cell constant checked as the sample entering the conductivity cell subsequently goes to drain and therefore does not compromise the water loop itself.

Figure 5. PAT700 direct conductivity analyser cell constant can be verified according to the requirements in USP<645>
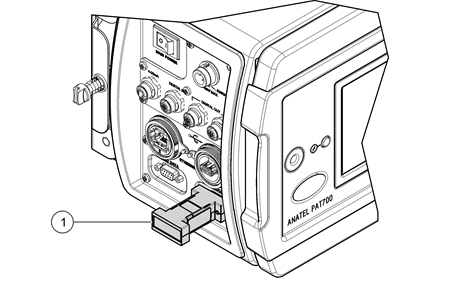
Figure 6. Beckman Coulter PAT700 direct conductivity analyser meter accuracy can be verified using external resistor as per the requirements of USP<645>
Excursion capture for root-cause analysis
TOC or conductivity contamination events can be transient in nature. For example, a water system that is experiencing gradual biofilm build up may experience TOC excursions just after sanitisation cycles as biofilm sloughs off the inside of the pipework. This excursion can quickly disappear, either by being diluted as it passes into the large quantity of water in the loop storage tank, or broken down by UV lamps connected to the pipework distribution network put in place to discourage microbial build-up. However, this rapid disappearance may mask the increasing biofilm build up until there is a breakdown of system control and run-away microbial contamination. The PAT700 can be programmed to capture a water sample should a TOC excursion be detected so that the sample can be further analysed to get to root cause.
Dual UV and UV detect
The primary method used by most pharmaceutical-grade TOC analysers to oxidise the TOC is UV light. Almost all TOC analysers using UV light only have a single UV lamp and do not monitor the level of UV light the lamp emits, thus potentially ignoring a reduction in UV light output that may compromise the analyser’s ability to measure TOC. Typical lifetime of these UV lamps is around 12 months and if the UV lamp fails completely, then the analyser will no-longer be able to analyse TOC. In modern WFI systems the level of TOC can be <10ppb and a failed TOC analysis UV lamp may go un-noticed, leaving potential TOC excursions in the WFI system to pass undetected. If a failure of the UV lamp is detected by the user, it may trigger the need for increased grab sample TOC analysis by the QC laboratory until the TOC analyser can get repaired. The PAT700 has main and a standby UV lights and monitors the UV light output. Should the UV light from the main lamp fall below an acceptable level for good TOC analysis, then the standby lamp is automatically activated and an alarm set to notify the user.
Figure 8. PAT700 ensures there is always sufficient oxidizing UV light by measuring UV output and having auto-switching main and standy-by UV lamps
Complete oxidation
The EP 2.2.44 chapter on TOC analysis emphasises the importance of complete oxidation of the organic contamination in order to get an accurate TOC measurement3. Longer chain, more complex organic molecules may create organic acids in the process of being oxidised down to CO2. Organic acids may contribute more strongly to the conductivity measurement than the final CO2 from the completely oxidised organic, so failure to ensure complete oxidisation of the organic may result in an incorrect TOC value. The PAT700 monitors the change in the conductivity measurement and waits until there is no more change in order to detect that the organic has been completely oxidised, thus ensuring accurate TOC analysis.
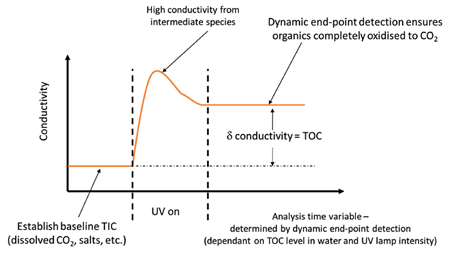
Figure 9. Beckman Coulter PAT700 uses dynamic end-point detection to ensure complete oxidation for accurate TOC analysis, even when UV lamp intensity decreases
TOC oxidisation curve profile
Some TOC analysers, such as the ANATEL PAT700 from Beckman Coulter, provide an indication of the oxidation curve profile during each TOC analysis. A change in the type of profile, combined with a change in TOC levels may provide insight into a potential degradation of the RO system integrity, prompting the user to investigate further and consider corrective action to prevent a large-scale contamination event and to put in place preventative measures to avoid the recurrence of the problem in the future.
Figure 10. Changes in TOC oxidation profile curve can indicate potential degradation of water treatment integrity, prompting investigation to prevent a large-scale contamination event
Conclusion
General purpose TOC analysers designed to fit many different types of application may not give the same level of confidence that analysers designed specifically for PW and WFI analysis in the regulated GMP environment such as the ANATEL PAT700 from Beckman Coulter. Manufacturers looking to take advantage of the new EP chapter allowing WFI production from RO treatment to reduce their overheads and carbon footprints may want a higher level of assurance that any slight change in TOC or conductivity will be detected accurately and robustly and may wish to consider selecting a TOC and conductivity analyser specifically designed for PW and WFI, rather than a general purpose TOC analyser.
References
- European Pharmacopeia (Ph. Eur.) Commission press release 18th March 2016 https://www.edqm.eu/sites/default/files/press_release_water_for_injections_march_2016_0.pdf
- EDQM Expert Workshop, 24 March 2011 European Directorate for the Quality of Medicines & Healthcare (EDQM) 7 allée Kastner, CS 30026 F -67081 Strasbourg
- US Pharmacopeia Convention, United States Pharmacopoeia, Rockville MD, USA and Council of Europe, European Directorate for the Quality of Medicines & Healthcare, European Pharmacopoeia, Strasbourg, France
- Council of Europe, European Directorate for the Quality of Medicines & Healthcare, European Pharmacopoeia 8.0, 01/2008:20244, Total Organic Carbon in Water for Pharmaceutical Use, Strasbourg, France
- International Society for Pharmaceutical Engineering, The ISPE Good Practice Guide: Ozone Sanitization of Pharmaceutical Water Systems, First edition July 2012 http://www.ispe.org/ispe-good-practice-guides/ozonesanitization- pharmaceutical-water-systems [14th August 2014]
- Pharmaceutical and Healthcare Sciences Society, Best Practice for Particle Monitoring

