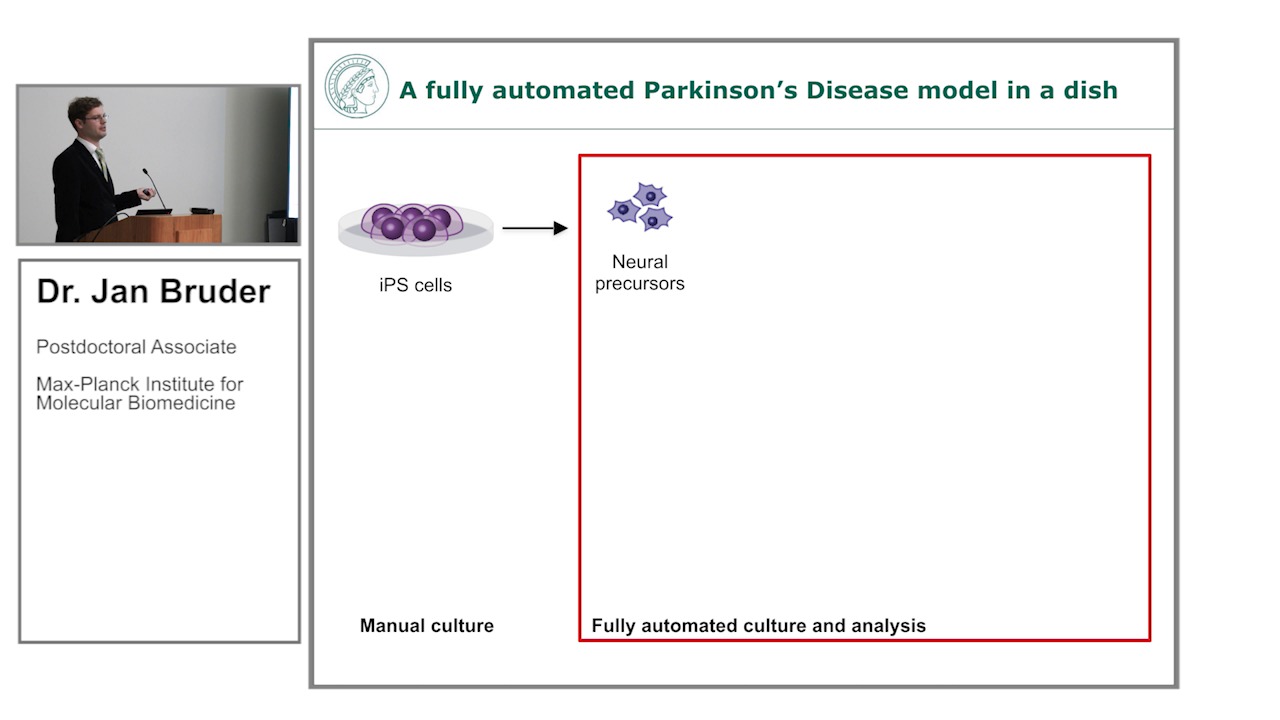
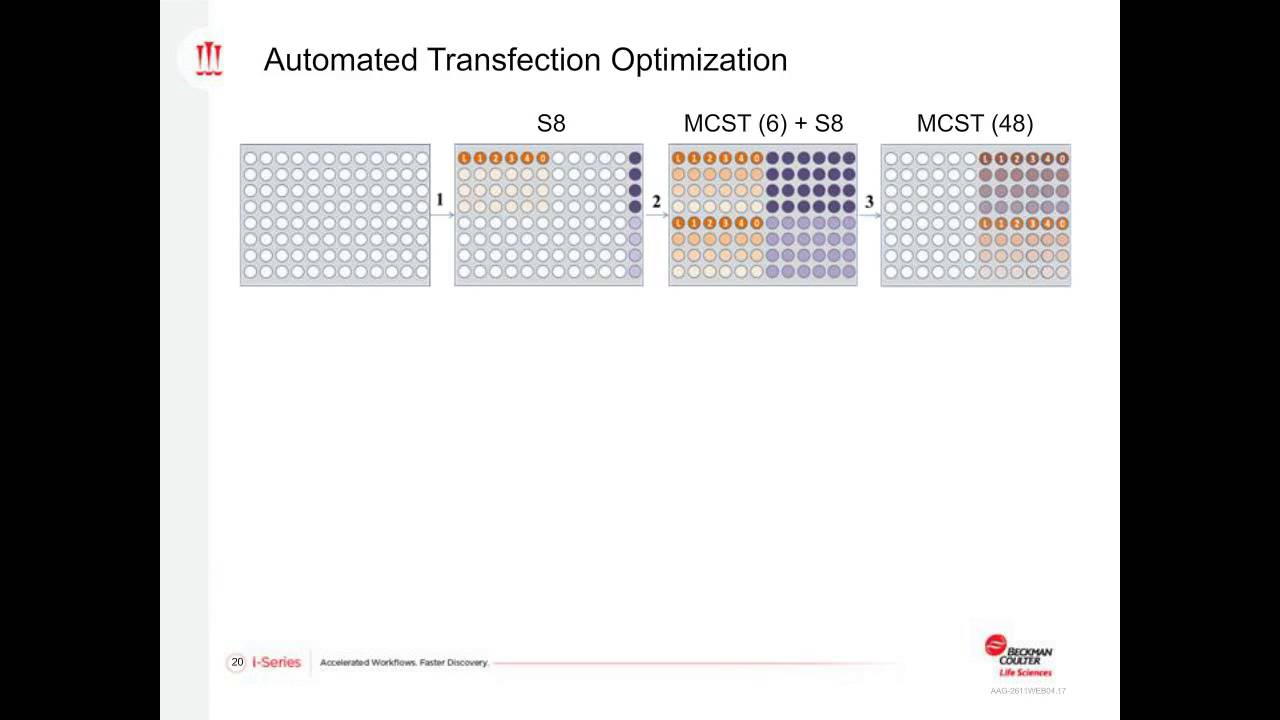
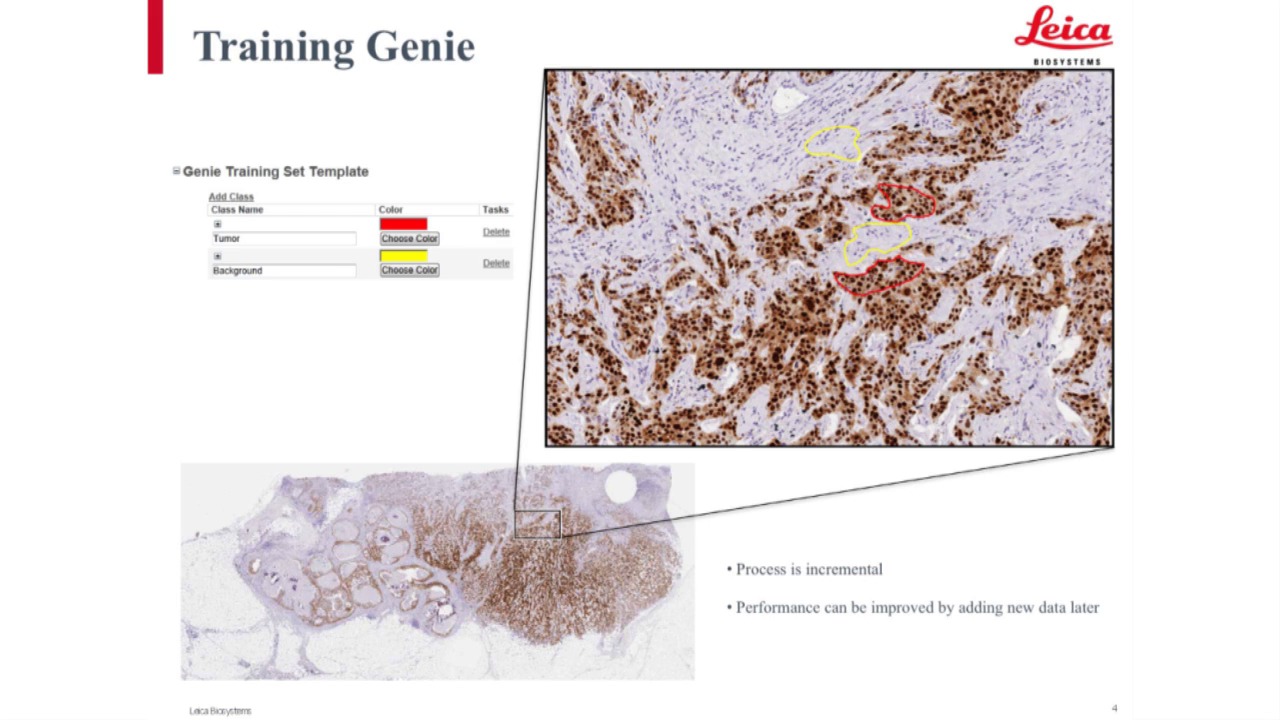
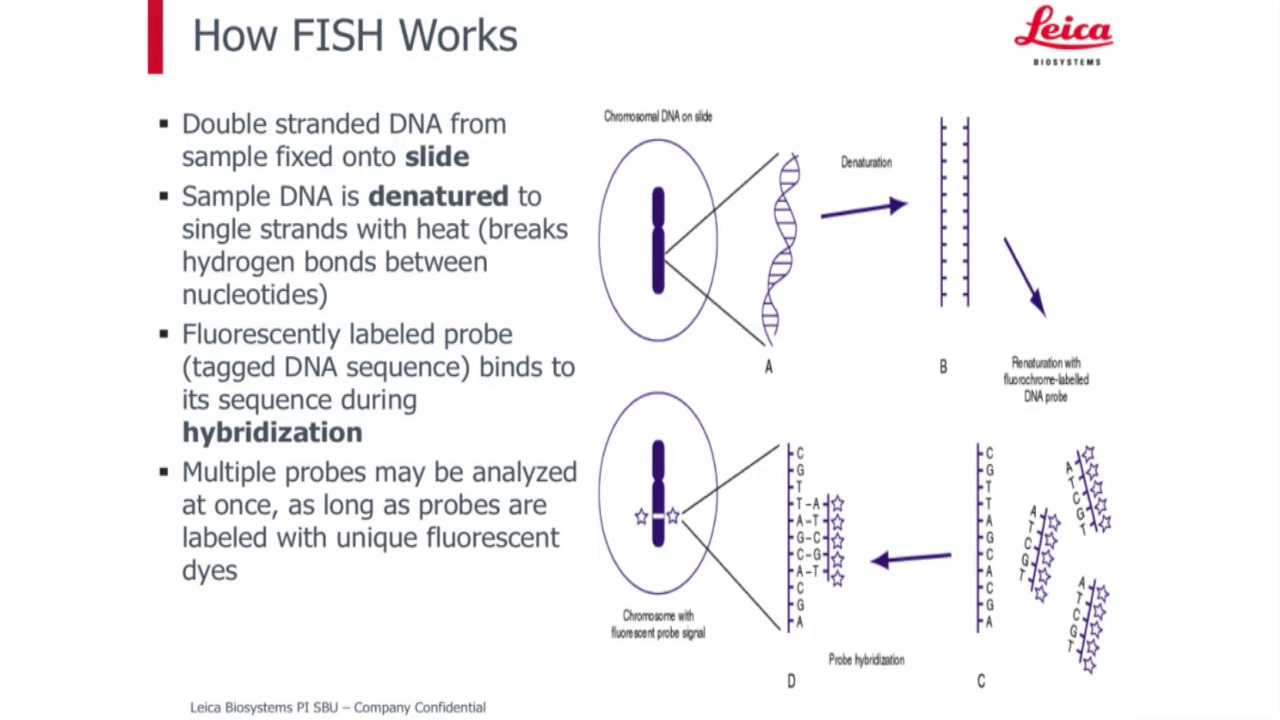
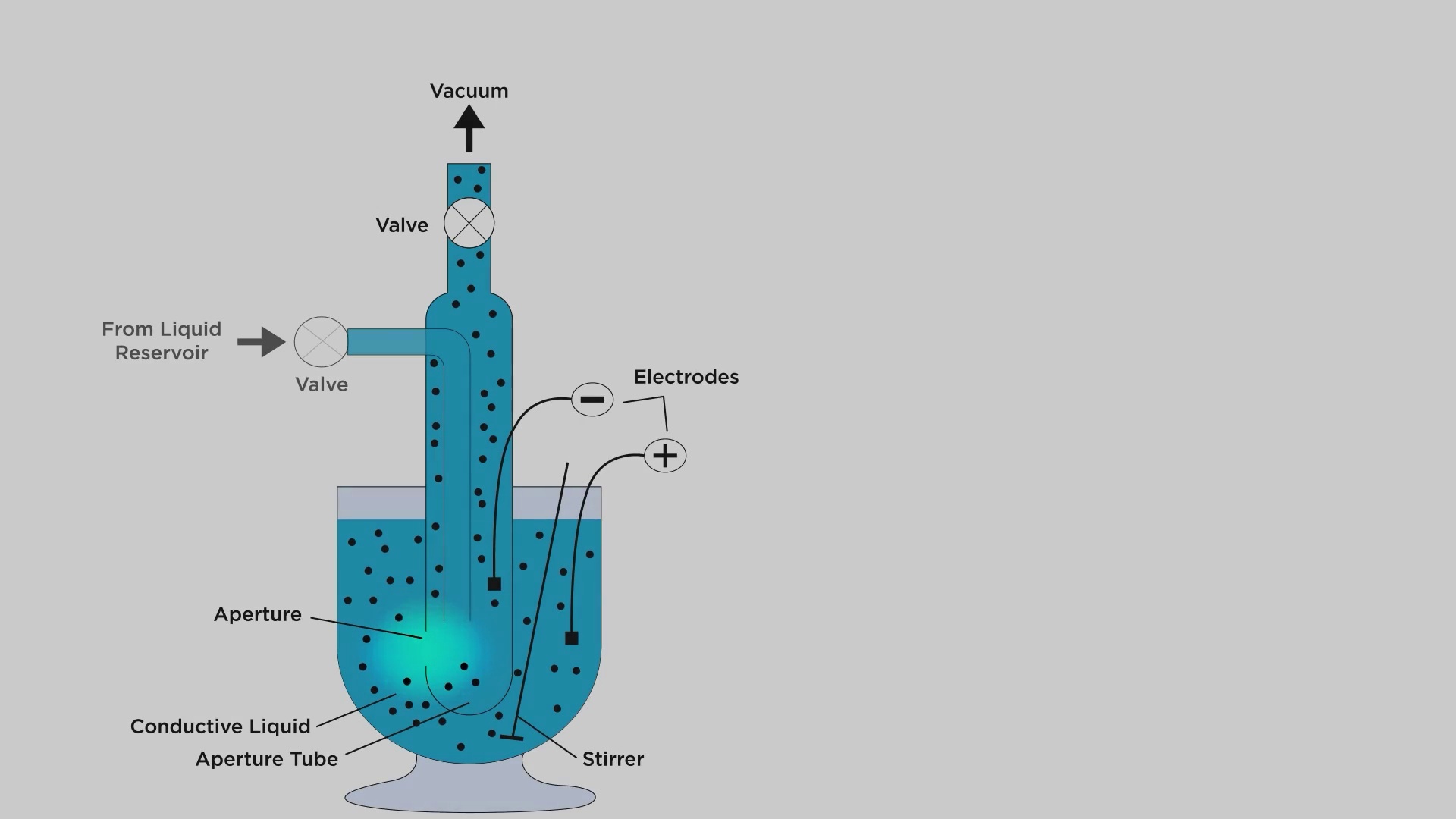
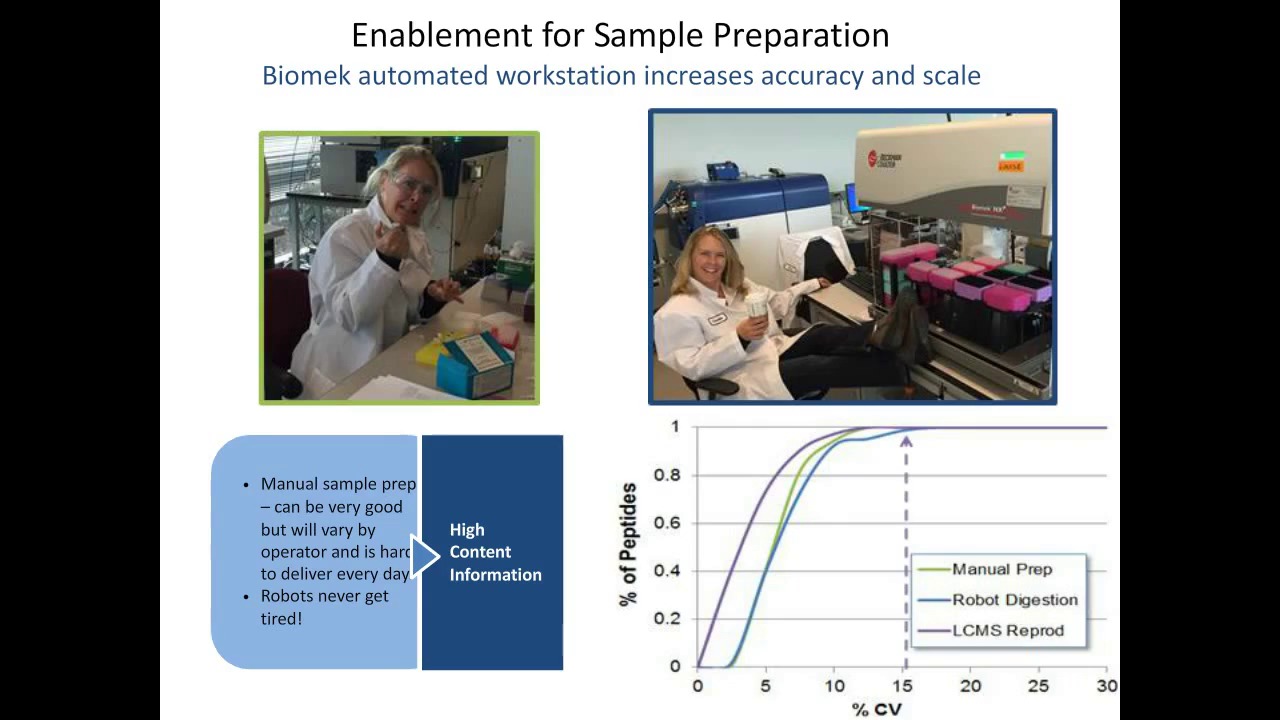
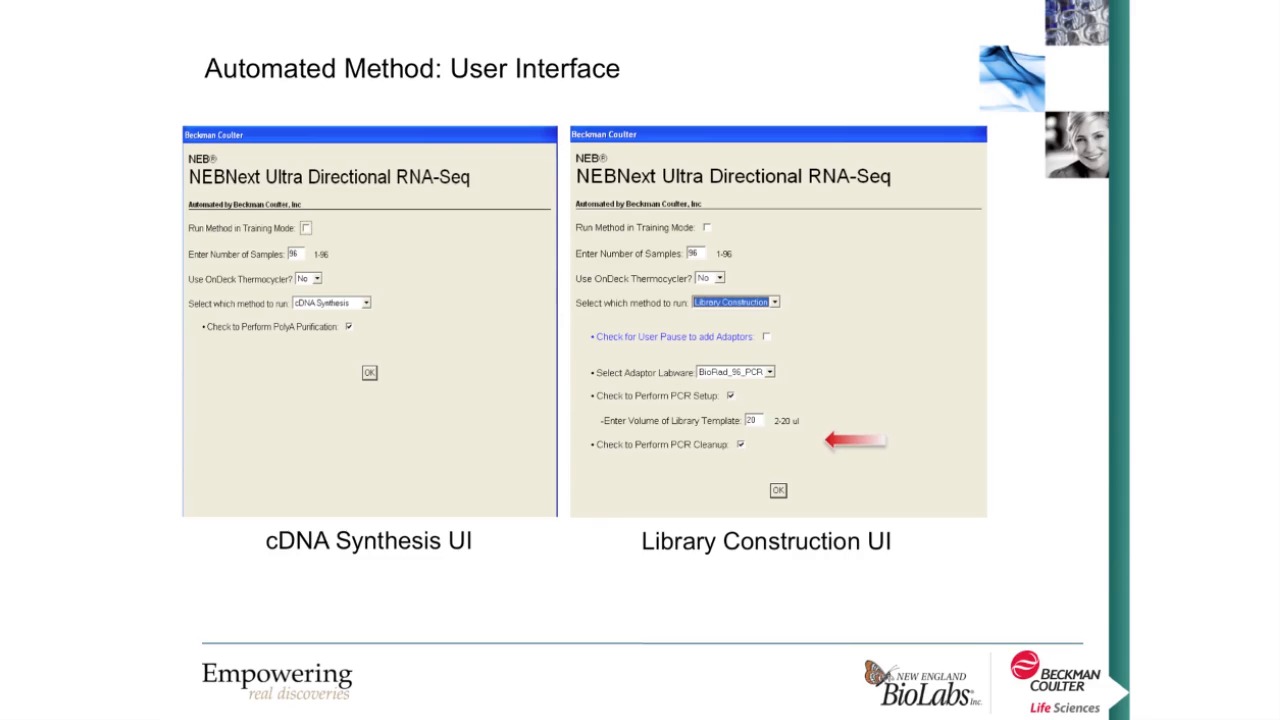
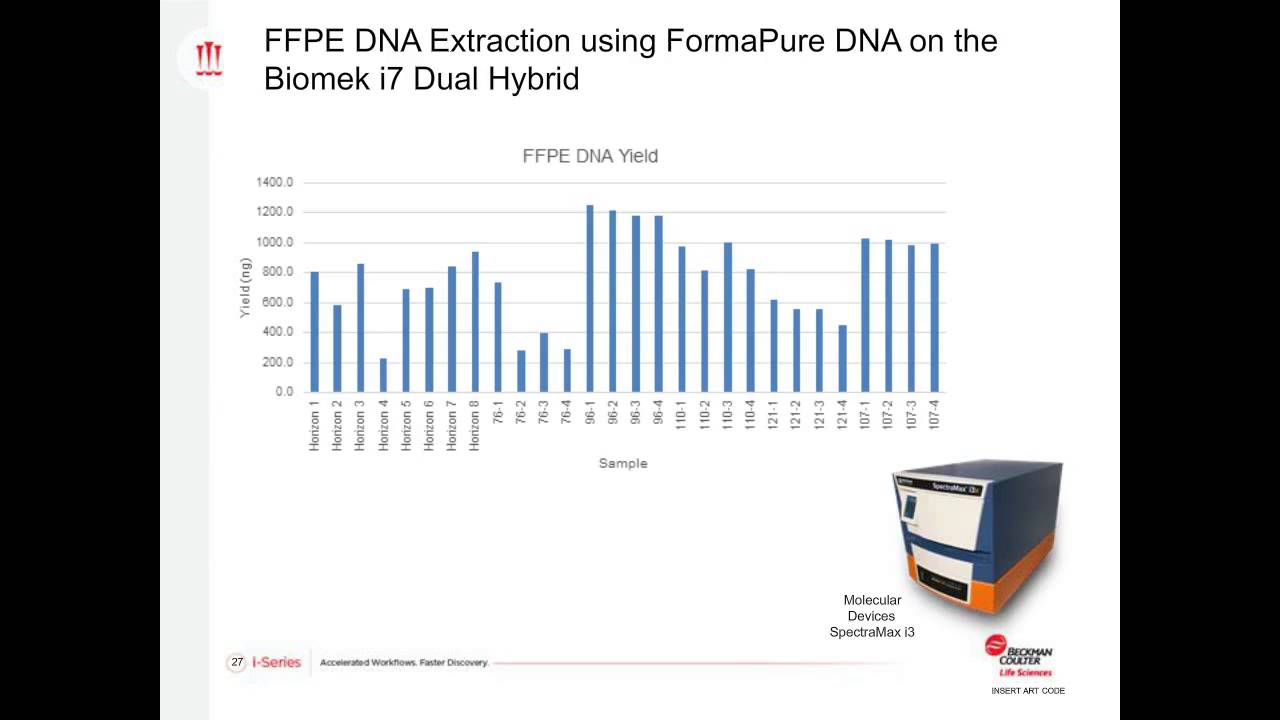
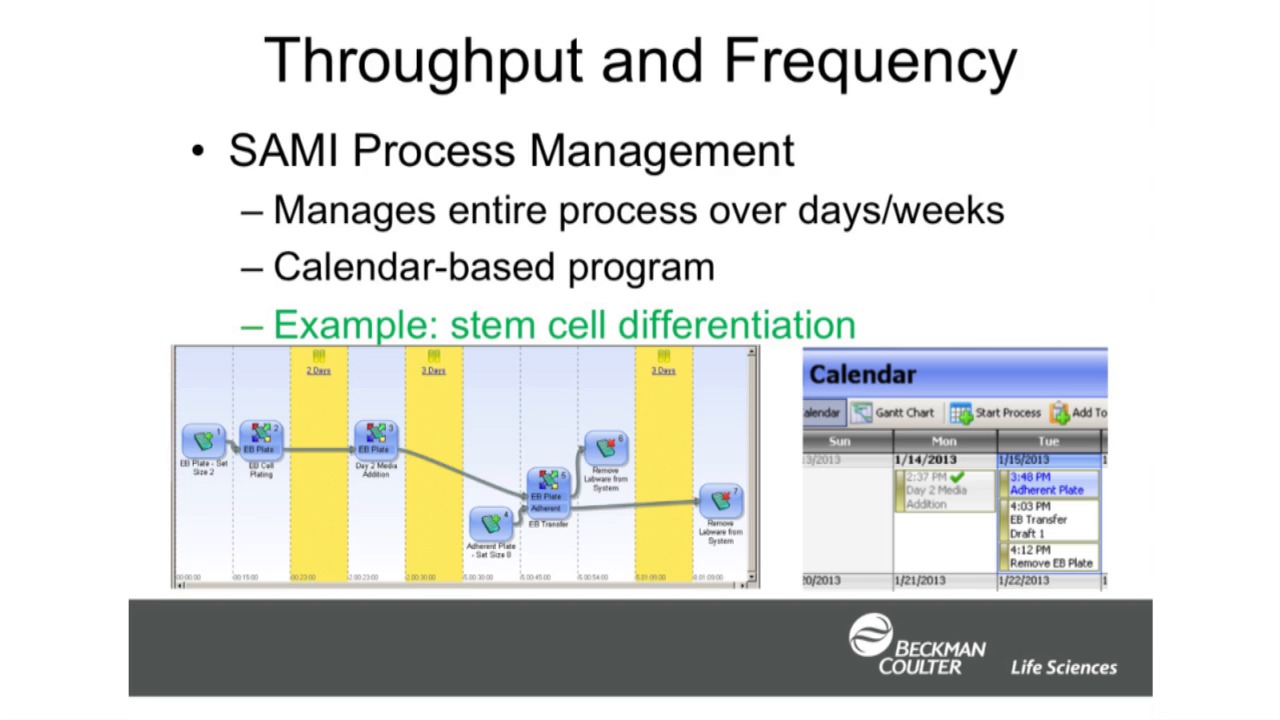
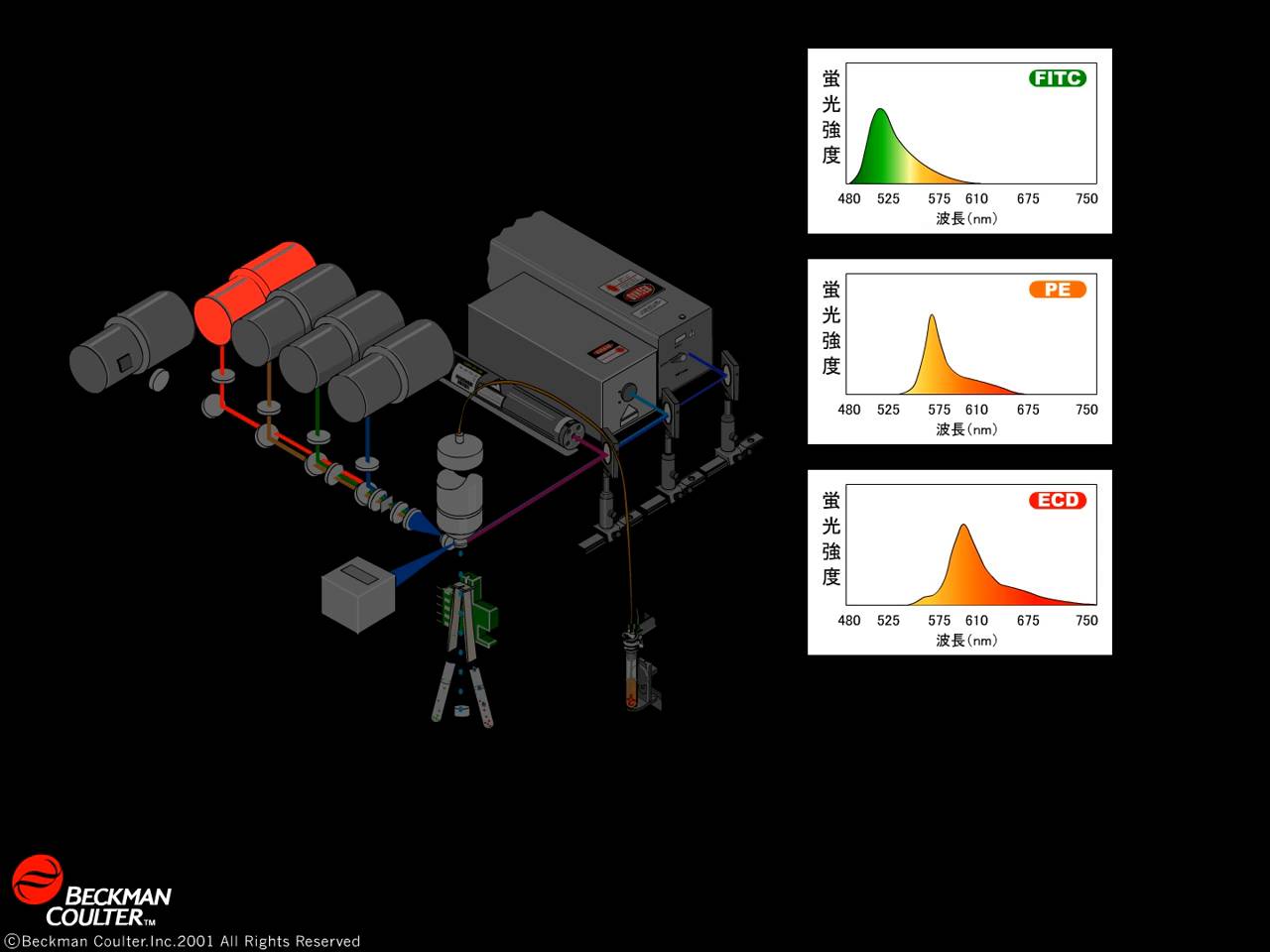
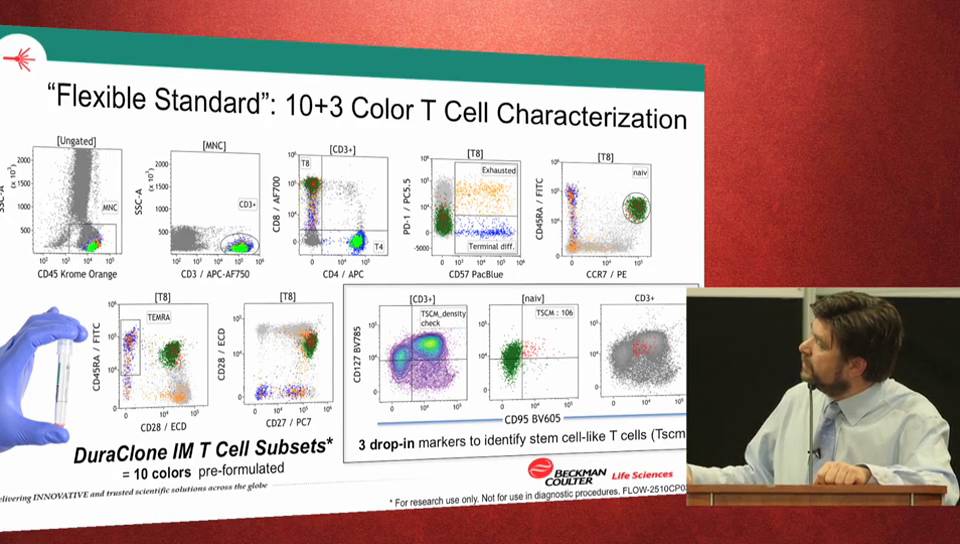
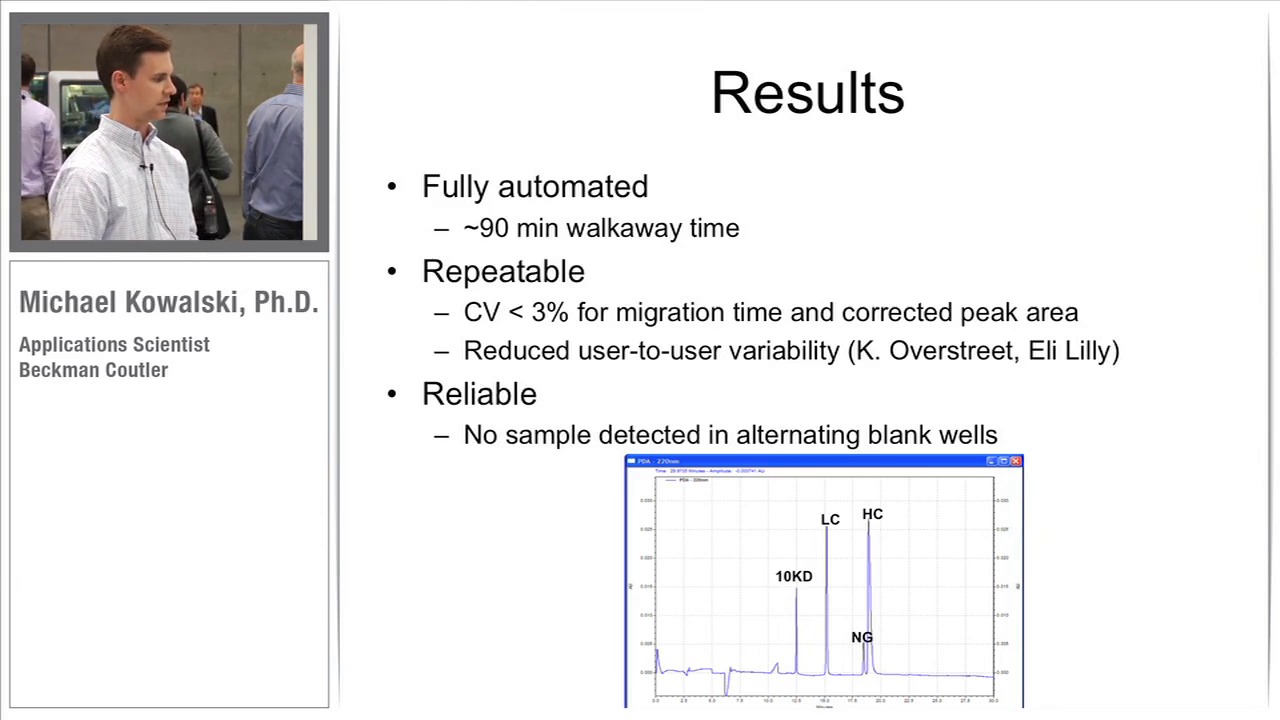
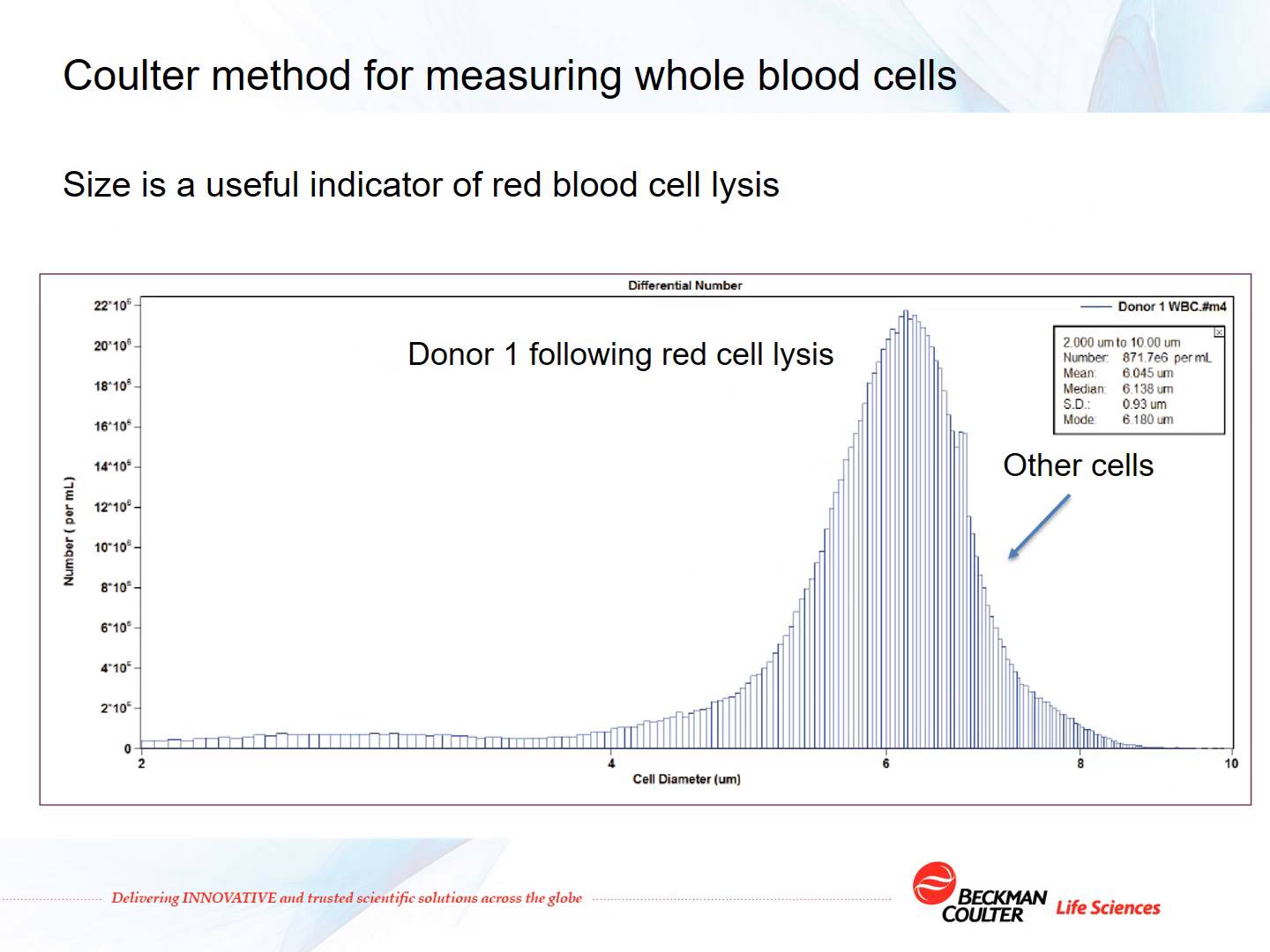
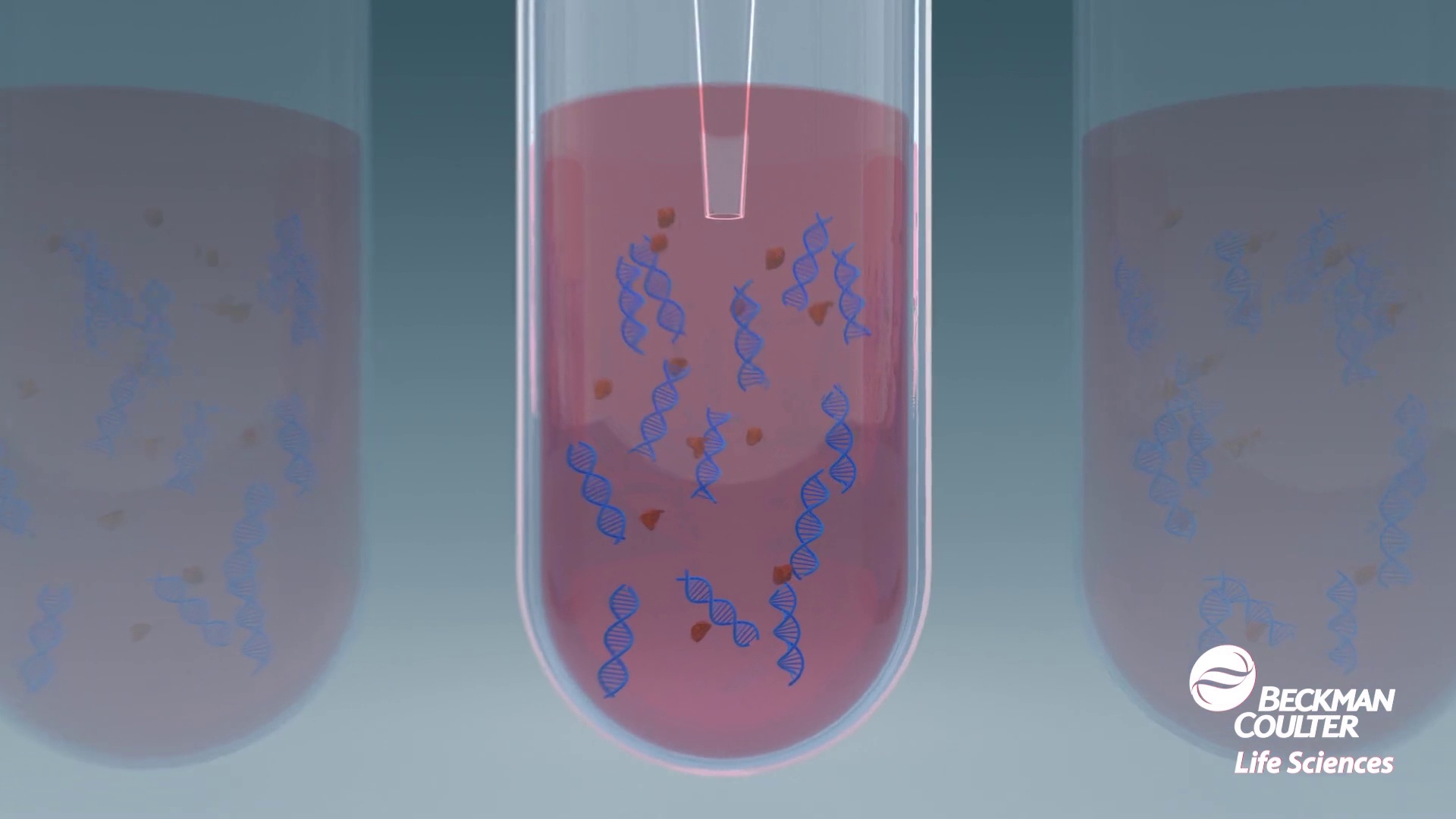
-
-
Melden Sie sich an, um die Vertragspreise anzuzeigen
Ihr Warenkorb ist leer
Einkaufswagen anzeigenMenge -
Bleiben Sie auf dem Laufenden mit unserem Newsletter.
Erhalten Sie aktuelle Informationen zu relevanten Themen von den Experten von Beckman Coulter Life Sciences. Geben Sie Ihre E-Mail ein.
© 2000-2026 Beckman Coulter, Inc. All rights reserved. Beckman Coulter, the stylized logo, and the Beckman Coulter product and service marks mentioned herein are trademarks or registered trademarks of Beckman Coulter, Inc. in the United States and other countries. All other trademarks are the property of their respective owners.

NOT ALL PRODUCTS ARE AVAILABLE IN ALL COUNTRIES. PRODUCT AVAILABILITY AND REGULATORY STATUS DEPENDS ON COUNTRY REGISTRATION PER APPLICABLE REGULATIONS The listed regulatory status for products correspond to one of the below: IVD: In Vitro Diagnostic Products. These products are labeled "For In Vitro Diagnostic Use." ASR: Analyte Specific Reagents. These reagents are labeled "Analyte Specific Reagent. Analytical and performance characteristics are not established." CE-IVD, CE: Products intended for in vitro diagnostic use and conforming to the In Vitro Diagnostic Regulation (IVDR) (EU) 2017/746. (Note: Devices may be CE marked to other directives.) RUO: Research Use Only. These products are labeled "For Research Use Only. Not for use in diagnostic procedures." LUO: Laboratory Use Only. These products are labeled "For Laboratory Use Only." No Regulatory Status: Non-Medical Device or non-regulated articles. Not for use in diagnostic or therapeutic procedures.