Arbeitsersparnis im Labor: manuelle und automatisierte Bearbeitung im Vergleich
Introduction
Flow cytometric immunophenotyping increasingly plays an integral role in the primary diagnosis and disease monitoring of many hematologic disorders. Accordingly, this has increased the need for consistent, high-quality testing methods. Advances in flow cytometry instrumentation, along with expanded availability of antibodies and fluorochromes, have improved identification of both normal and aberrant cell populations. However, automation in flow cytometry has not advanced as quickly as it has in other areas of the lab, and flow cytometry sample preparation remains a primarily manual process requiring highly skilled laboratory technologists.
Automation of sample processing presents a significant opportunity to address some of the challenges clinical flow cytometry laboratories face — with increasing test volume despite static or decreasing budget and staffing levels — while also improving quality, and reducing the potential for errors that can arise from manual methods.
The CellMek SPS Sample Preparation System can automate cell washing, antibody staining, lysing, and fixing samples for flow cytometry without any user intervention. It provides on-demand processing by running multiple programmable preparation methods in parallel, and continuous output of ready-to-analyze samples.
About the Testing Site
Tampa General Hospital is a 1041-bed hospital that serves as a regional safety net and the primary teaching hospital for USF Morsani College of Medicine.
The Flow Cytometry Lab is part of the Esoteric Testing Lab and performs over 2700 leukemia and lymphoma immunophenotyping analyses per year, in addition to lymphocyte subset quantitation, paroxysmal nocturnal hemoglobinuria (PNH) evaluations, fetal hemoglobin evaluations, and other specialized molecular and immunology testing.
Challenges
Coping with increased caseloads despite static or decreasing budgets and staffing levels
Flow cytometry labs are pressured to provide fast and accurate test results that enable clinicians to treat patients appropriately, while also meeting administrative expectations for productivity, staffing and training. This drives a continuous need for optimized workflows.
The US vacancy rate for highly trained flow cytometry technologists is approaching 10 percent. Workload from vacant positions is taken on by existing staff, increasing exhaustion and burnout, and in some cases limiting testing capacity.
Despite recent improvements to the flow cytometry ordering process and workflow, flow cytometry technologists spend significant time manually preparing each sample in addition to analyzing results. Automation of sample processing helps to reduce workloads and get test results to patients more quickly, thereby expediting their access to vital care.
Standardizing processing for reproducible results
Manual sample processing is dependent on each technologist’s unique skill level and technique, introducing an element of variability that can be difficult to differentiate from other sample-specific sources of variance. Variability, such as incomplete lysis and pipetting errors, introduces the potential for inconsistent results.
Automating manual steps in sample processing creates standardization and brings consistency to the process, improving workflow and reducing potential sources of error.
Customized Assays
Customized assays present challenges for automation because testing protocols and characteristics are determined by the individual lab and not by the manufacturer. Often, laboratories utilize multiple manufacturer’s antibodies and different antibody panel combinations, as well as variable processing and staining procedures to obtain test results.
The CellMek SPS sample preparation system is fully programmable, and capable of accommodating user-defined protocols, reagents, and antibodies to fully automate each lab’s unique processes.
Automating a customized assay
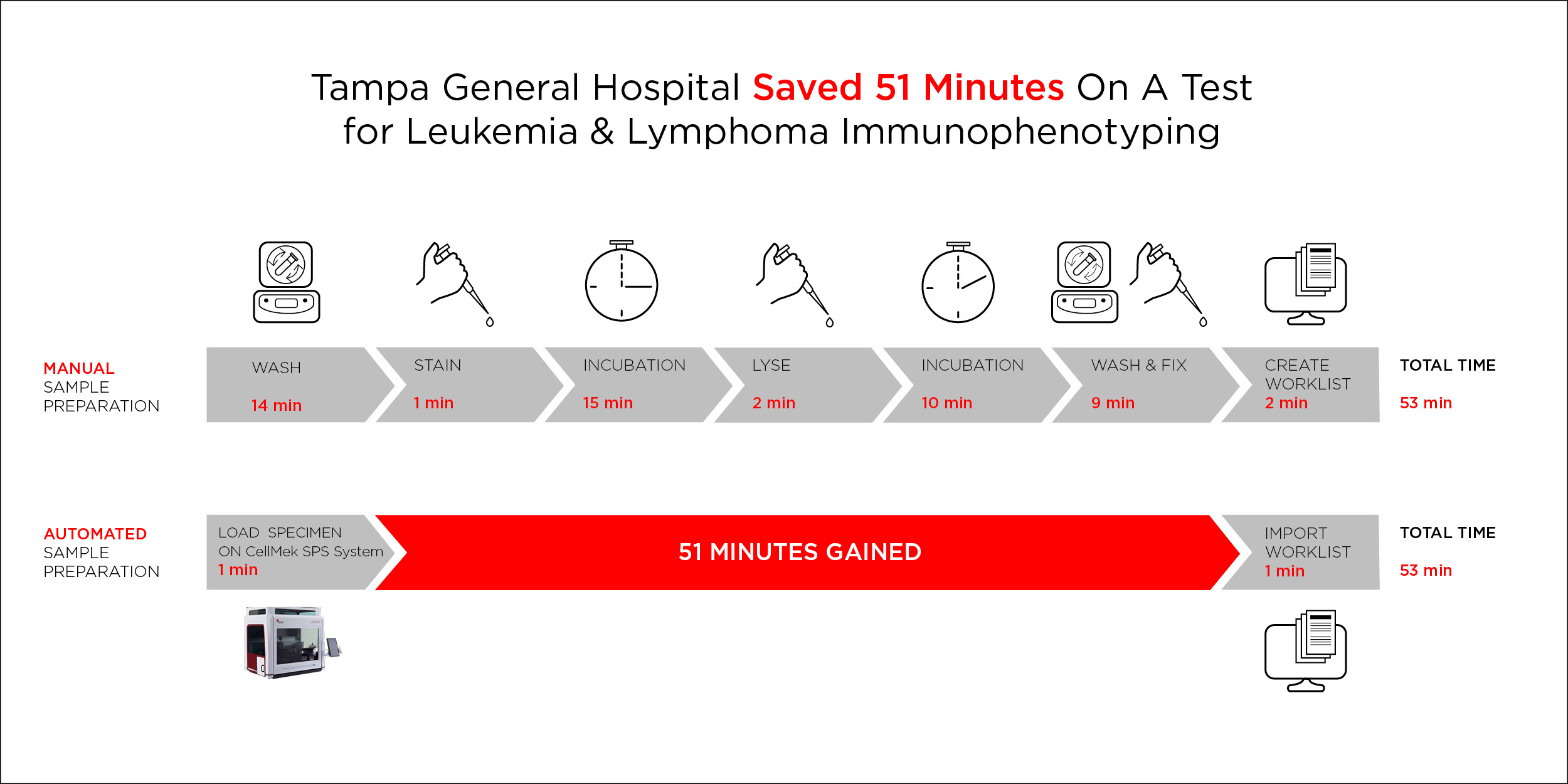
Sample processing for a leukemia and lymphoma (L&L) immunophenotyping assay was automated on the CellMek SPS Sample Preparation System using the CellMek Panel Designer Software. Incubation times were defined and optimized based on the specific procedure, and custom reagent labels were used to accommodate reagents and antibodies not manufactured by Beckman Coulter. Specimen washing was set to 5 cycles, 2% FCS in PBS was used in the wash buffer to address nonspecific binding, and 3X fix was made to account for differences between the automated protocol and the manual test.
Hands-on time for the purpose of this study includes technologist interactions and process monitoring.
Figure 1. Reduction in error-prone steps and hands-on time for leukemia and lymphoma Immunophenotyping sample processing with CellMek SPS Sample Preparation System.
*Spin time included
Figure 2. Increased efficiency with CellMek SPS System for leukemia and lymphoma immunophenotyping.
As minimum incubation times are fixed, processing times on the CellMek SPS System cannot be significantly faster than manual sample preparation. However, sample preparation with the CellMek SPS System yields significant savings in hands-on time, as the process is fully automated, requiring minimal input from the technologist. Automation on the CellMek SPS System reduced the hands-on time for sample preparation of peripheral blood for L&L immunophenotyping assay from 28 minutes to 2 minutes, thereby increasing the time available for analysis or other tasks by 118%.
Discussion
Overall, we found automation of our assays on the CellMek SPS Sample Preparation System to be a straightforward process that resulted in significant savings in technologist hands-on time. Automated preparation of flow cytometry samples presents a significant opportunity to streamline, optimize, and standardize workflows in flow cytometry laboratories.
References
- Garcia E, Kundu I, Kelly M, Soles R. The American Society for Clinical Pathology 2020 Vacancy Survey of Medical Laboratories in the United States. Am J Clin Pathol. 2021 Dec 1; aqab197. doi: 10.1093/ajcp/aqab197.
- Brestoff JR, Frater JL. Contemporary Challenges in Clinical Flow Cytometry: Small Samples, Big Data, Little Time. J Appl Lab Med. 2022 Jun 30;7(4):931-944. doi: 10.1093/jalm/jfab176. PMID: 35061902.
- Fiona E. Craig, Kenneth A. Foon; Flow cytometric immunophenotyping for hematologic neoplasms. Blood 2008; 111 (8): 3941–3967. doi: https://doi.org/10.1182/blood-2007-11-120535
Results may vary depending on user factors, including protocol and assay being performed. The results are based on an efficiency study completed by Tampa General Hospital, USA, wherein one person was dedicated to either automated or manual preparation.
Marci O’Driscoll, MS, MT
Marci received her bachelor’s degree in Interdisciplinary Natural Science, followed by her Master's degree in Chemistry from the University of South Florida. Shortly after, she began her research career at the Children’s Research Institute. Working in various labs, she became proficient in designing, implementing, and troubleshooting experiments, using biological, molecular, and flow cytometry methods. During this time, she discovered her passion for bringing research from the bench to the bedside and completed her clinical laboratory training at Johns Hopkins All Children’s Hospital. She currently combines her love for clinical diagnostics, research, and teaching as a Clinical Scientist, responsible for the evaluation and validation of new flow cytometry testing in the Esoteric Lab at Tampa General Hospital.